With ISO 14644 in a state of flux, cleanroom users need to stay informed and contribute to the development of all parts of the standard. Jane Ellis attended the BSI’s recent Cleanrooms Consultation seminar to hear views about the latest changes and their impact.
The BSI’s Cleanrooms Consultation meeting, held in London in March, provided an update to the revisions that have been made to the ISO/Draft International Standard (DIS 14644-1) for cleanrooms and it allowed delegates to voice their opinions and concerns about the proposed changes.
The BSI meeting was once again chaired by expert Gordon Farquharson, of Critical Systems Ltd. As chairman of the BSI LBI/030 committee responsible for the UK input to cleanroom standards, as well as chairman of CEN TC243 (the European committee for cleanroom standards) and convenor of ISO TC209 WG1, the international working group looking at airborne particulate cleanliness classes, Farquharson is well placed to inform on the recent changes to ISO 14644 and to outline the changing relationship of GMP guidance with ISO 14644-1.
Tony Harrison, life sciences manager at Pharmagraph, vice chairman of the UK PHSS Society and technical expert on BSI LBI/30, examined the impact of the normative reference to ISO 21501 Determination of particle size distribution, which now makes compliance with ISO 21501-4 a requirement. ISO 21501-4 provides a calibration procedure and verification method for particle counters used to classify and monitor cleanrooms.
Close to publication
Work on revising ISO 14644-1 Classification of air cleanliness by particle concentration and 14644-2 Specifications for testing and monitoring to prove continued compliance with ISO 14644-1 started back in 2005 and the revised standards should be published by the end of this year or early in 2012.
The Draft International Standard (DIS) enquiry and vote for these standards closed on 2 May. The opinions voiced at the BSI meeting contributed to the UK comments on the revision submitted in April, said Farquharson.
The revision of ISO 14644-1:1999 was triggered, he says, because users wanted an easier tool for calculating cleanroom classifications and felt that the current statistical method did not work as intended.
The nine ISO cleanliness classes will be unchanged, but a new table (see Table 1) defines the particle concentration at various particle sizes for each class.
| Table 1: Air cleanliness classification table by particle concentration | ||||||
| Maximum allowable concentration (particles/m3) for particles equal to and greater than the considered sizes shown belowa | ||||||
| ISO classification number N | 0.1µm | 0.2µm | 0.3µm | 0.5µm | 1µm | 5µm |
| ISO Class 1 | 10b | d | d | d | d | e |
| ISO Class 2 | 100 | 24b | 10b | d | d | e |
| ISO Class 3 | 1000 | 237 | 102 | 35b | d | e |
| ISO Class 4 | 10,000 | 2,370 | 1,020 | 352 | 83b | e |
| ISO Class 5 | 100,000 | 23,700 | 10,200 | 3,520 | 832 | e |
| ISO Class 6 | 1,000,000 | 23,7000 | 102,000 | 35,200 | 8,320 | 293 |
| ISO Class 7 | c | c | c | 352,000 | 83,200 | 2,930 |
| ISO Class 8 | c | c | c | 3,520,000 | 8,320,000 | 29,300 |
| ISO Class 9 | c | c | c | 35,200,000 | 8,320,000 | 293,000 |
a. All concentrations in the table are cumulative, e.g. for ISO Class 5, the 10,200 particles shown at 0.3µm include all particles equal to and greater than this size.
b. These concentrations will lead to large air sample volumes for classification. Sequential sampling procedure may be applied; see Annex D.
c. Concentration limits are not applicable in this region of the table due to very high particle concentration.
d. Sampling and statistical limitations for particles in low concentrations make classification inappropriate.
e. Sample collection limitations for both particles in low concentrations and sizes greater than 1µm make classification inappropriate, due to potential particle losses in the sampling system.
The use of Table 1 will ensure a better definition of the appropriate particle-size ranges for the different classes. However, the formula (see below) will be retained to allow definition of intermediate decimal classes:
The standard retains the concept of ultrafine and macroparticle descriptors for particle sizes outside the range appropriate for measurement using airborne particle counters.
The consensus at the BSI meeting was that this change would be unlikely to have a major impact on business and could help prevent poor selection by companies in terms of facility size versus class.
Users of cleanrooms also wanted a more logical selection of the number of sample locations and a simplification of the statistical approach using the 95% UCL (Upper Confidence Level) evaluation of class compliance for 2–9 locations in ISO 14664-1:1999.
The major problem with the approach suggested, said Farquharson, is that it assumes an even distribution of contamination in a cleanroom, which is often not the case, particularly in non-directional cleanrooms during operation.
It is therefore proposed to change this to a table of sample locations, based on ensuring 95% level of confidence that at least 90% of the cleanroom or clean zone will comply with the maximum particle concentration limit for the target class of air cleanliness.
This will make no change to the basic mechanics of classification, nor does it affect real-time monitoring, but it would require a new classification of existing areas, said Farquharson (see Table 2).
| Table 2: Proposed new classification procedure in ISO 14644-1: 2010 | |
| Area (m2) less than or equal to | Min. number of sample locations (NL) |
1. Determine the number of sample locations (NL) from table
2. Divide the room into NL sub-regions of the same area and measure the particle concentration at a randomly selected location within each sub-region
3. If all NL measured particle concentrations comply with the class limit, the classification is passed
A new principle for selection and acceptance of the sample locations has also been incorporated into the revised ISO/DIS 14644-1, which says the measurement of locations must be selected randomly in the cleanroom or zone and that new locations must be selected at every re-classification. ISO 14644-1:1999 did not state how to select locations, but it is common practice to select them on a regular grid across the room.
According to the regulators, the advantage of a randomised sampling plan is that all parts of the room have the same chance of being measured. It also allows companies to quantify and control, in a statistical sense, the risk induced by measuring at only a limited number of locations, and it ensures that the results can be reliably generalised for the entire room.
The number of sample locations has increased with the revision, thus requiring more measurement efforts (see Figure 1).
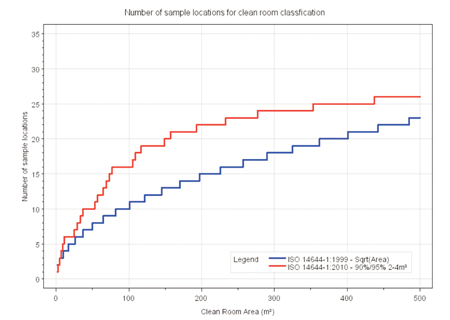
Figure 1: Comparison of the number of sample locations for cleanroom classification by the old (1999) and new (2010) version of ISO 14644-1.
Source: Niels Vaever Hartvig et al, Journal of the IEST, 54, 1, Special ISO Issue
Tim Triggs, director of filter testing company DOP Solutions, of Letchworth, Hertfordshire, UK, said revising the number of sample locations would place a much greater administrative burden on businesses, while DOP’s technical director Neil Stephenson said it would mean that testing would take a third longer than previously.
Along with many other delegates, Stephenson also questioned the randomised sampling plan, stating that it would not allow comparisons to be made between the results from one year to the next, which is vital when a contamination problem is encountered.
Risk-based decisions
A simplification of the table of test frequencies is the main proposed change to ISO 14664-2:2000. Re-testing will be required annually, but this annual interval can be extended if real-time monitoring says the room/device is in compliance (pressure and airborne particles).
Farquharson said this will make it easier to use risk-based decision making about testing frequency. It also reinforces that ‘Demonstration of continued compliance with ISO 14644-1’ requires a documented monitoring and periodic test plan; the undertaking of monitoring and periodic testing; and acting on the results. In non-regulated cleanroom sectors it provides a minimum best practice.
New informative sections will also be required in ISO 14644-2 to specify the essential requirements of real-time monitoring systems, covering particle, pressure difference and air velocity/volume.
With the revision of ISO 14644-1, the regulators are now taking more interest in ISO 21501-4:2007. The calibration standard has been in print for three years, and it describes a calibration and verification method for a light scattering airborne particle counter, which is used to measure the size and particle number concentration of particles suspended in air. The light scattering method is based on single particle measurements. The typical size range of particles measured by this method is between 0.1µm and 10µm.
Instruments that conform to ISO 21501-4:2007 are used for the classification of air cleanliness in cleanrooms and associated controlled environments in accordance with ISO 14644-1, as well as the measurement of number and size distribution of particles in various environments.
According to Tony Harrison, ISO 21501 has remained a ‘best-kept secret’ for many years, and if it is to become a normative reference, all particle counters would have to be re-calibrated to comply with it.
DOP Solutions’ Triggs expressed the view that ISO 21501 should not become a normative reference, as it would be too costly for suppliers of particle counters.
Stephenson added: “It’s a great standard, but to insist that five-year-old particle counters should be thrown away is nonsense.” He also questioned whether it would be achievable to reconfigure every particle counter in the field.
BSI’s programme manager and secretariat of CEN/TC 352, David Michael, said he was not opposed to the inclusion of ISO 25101 for reference, but questions remained about older particle counters and how they complied with the standard and how it could be enforced. He suggested that the regulators should specify a time range for compliance.
Preliminary work has also begun on revising the ISO 14698: Classification of airborne biocontamination in cleanrooms, and Classification of surface biocontamination, including methods of measurement and their validation. Harrison explained that the methods of measurement and their validation might be replaced or become part of the new ISO 14644 standard.
GMP impact
When summing up the impact of the changes to ISO 14644-1 on pharmaceutical GMPs, Farquharson described the GMPs and the 1999 version of ISO 14664-1 as “joined at the hip”, but said there is a problem with regulatory alignment in that the US FDA 2004 Aseptic Processing Guidance differs from the EU, PIC/S, WHO, China S/FDA model (see Table 3).
| Table 3: Air Classifications/US FDA 2004 Aseptic Processing Guidance | |||||
| Clean area classification (0.5µm particles/ft3) | ISO designationb | >0.5µm particles/m3 | Microbiological active air action levelsc (cfu/m3) | Microbiological plate action levels (dia 90mm cfu/4hrs) | |
| 100 | 5 | 3,520 | 1e | 1e | |
| 1000 | 6 | 35,200 | 7 | 3 | |
| 10,000 | 7 | 352,000 | 10 | 5 | |
| 100,000 | 8 | 3,520,000 | 100 | 50 | |
a. All classifications based on data measured in the vicinity of exposed materials/articles during period of activity
b. ISO 14644-1 designations provide uniform particle concentration values for cleanrooms in multiple industries. An ISO 5 particle concentration is equal to Class 100 and approximately equals EU Grade A
c. Values represent recommended levels of environmental quality. You may find it appropriate to establish alternate microbiological action levels due to the nature of the operation or method of analysis.
d. The additional use of settling plates is optional
e. Samples from Class 100 (ISO 5) environments should normally yield no microbiological contaminants
All air classifications in the US are based on data in the vicinity of exposed materials during periods of activity and only one particle size – 0.5µm – is considered. The US also says the additional use of settle plates with an air sampler is optional and ISO 5 cleanrooms and higher should not consider >5µm particles because they normally ‘yield no microbiological contaminants’.
In the rest of the world, rest periods and periods of activity are considered and two particle sizes of 0.5µm and 5µm are sampled.
According to Harrison, monitoring the >5.0µm particle concentration count is an important diagnostic tool for early detection of failure. The consecutive or regular counting of low levels of >5.0µm particles is an indicator of possible contamination and should be investigated. Particles of this size could be skin particles carrying microbial contamination, he said.
Harrison also believes that settle plates have a very important role to play and he gave an example of a series of tests performed in a fully functional cleanroom to establish contamination effects.1 Five tests took place when the cleanroom was empty, when scientists were fully gowned, partially gowned, in everyday office clothes and when jogging in shorts and T-shirts.
“The tests showed that the contamination source was people, not failing HEPA,” said Harrison. “A cleanroom can be out of compliance at >5µm while still complying at >0.5µm.”
The removal of ISO Class 5 5µm particles from the proposed ISO 14644-1 Classification Table was a contentious issue at the BSI meeting. Bob Somerville, partner at Robert Somerville Associates, a consulting engineering partnership based in Watford, UK, suggested that the new proposal should include a risk assessment depending on use and a clear majority, particularly those operating in the life sciences, asked for the 5µm particle limit to remain.
Future work
The industry now almost has a complete set of standards that allow cleanrooms to be classified for cleanliness by airborne particles, surface particles, airborne chemicals and surface chemicals. Standards for airborne and surface microbiological organisms are in progress.
The ISO TC 209 is starting to look at process impact with/on cleanrooms and nanoparticle requirements, while BSI LBI/030 is working on a cleanroom energy Code of Practice (BS 8568), which supports the energy standard BS EN16001. CT
Reference
1. John Sharp, Adam Bird, Sebastian Brzozowski and Kay O’Hagan. European Journal of Parenteral & Pharmaceutical Sciences 2010; 15(3): 5–11




